System Industrie Electronic Deutschland GmbH
+49 87 1965 7807 0+49 87 1965 7807 20
solutions@sie.at
We are your partner for the development and production of embedded systems and cyber physical systems with a focus on real added value for people.
read moreOver 25 years of experience have allowed us to gather important expert know-how as a company in various industries. Click on your industry to learn more about our industry-specific specializations and process know-how.
As specialists in the development and production of embedded systems for the medical environment, we understand the special requirements of this complex and normatively demanding market.
read more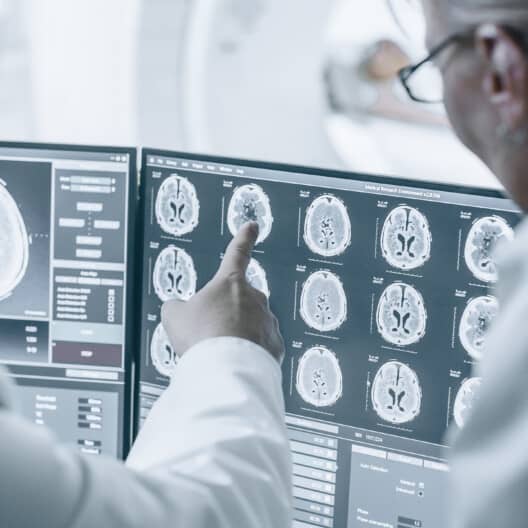
Our common focus as a development and manufacturing partner in the context of industrial embedded systems is therefore the flexible development and optimization of complete systems based on field-proven building blocks.
read more
Together with our strategic partner – secunet Security Networks AG – we have been active in the field of cybersecurity solutions for many years.
read more





We, the S.I.E team, are convinced that digital solutions must always focus on people and their needs.
Therefore, everything we do serves to sustainably generate real added value along entire value chains, all the way to the end users of digital products and services.
Together, we realize our vision of a human-centric digitization.

Zum Absenden des Formulars muss Google reCAPTCHA geladen werden.
Google reCAPTCHA Datenschutzerklärung
Zum Absenden des Formulars muss Google reCAPTCHA geladen werden.
Google reCAPTCHA Datenschutzerklärung
Zum Absenden des Formulars muss Google reCAPTCHA geladen werden.
Google reCAPTCHA Datenschutzerklärung
Zum Absenden des Formulars muss Google reCAPTCHA geladen werden.
Google reCAPTCHA Datenschutzerklärung